News
Perfusion Cultivation of Artificial Liver Extracellular Matrix in Fibrous Polymer Sponges Biomimicking Scaffolds for Tissue Engineering
11.08.2020
Michael Mader, Moritz Helm, Mingxia Lu, Martina H. Stenzel, Valérie Jérôme, Ruth Freitag, Seema Agarwal, Andreas Greiner
Biomacromolecules 2020, https://doi.org/10.1021/acs.biomac.0c00900
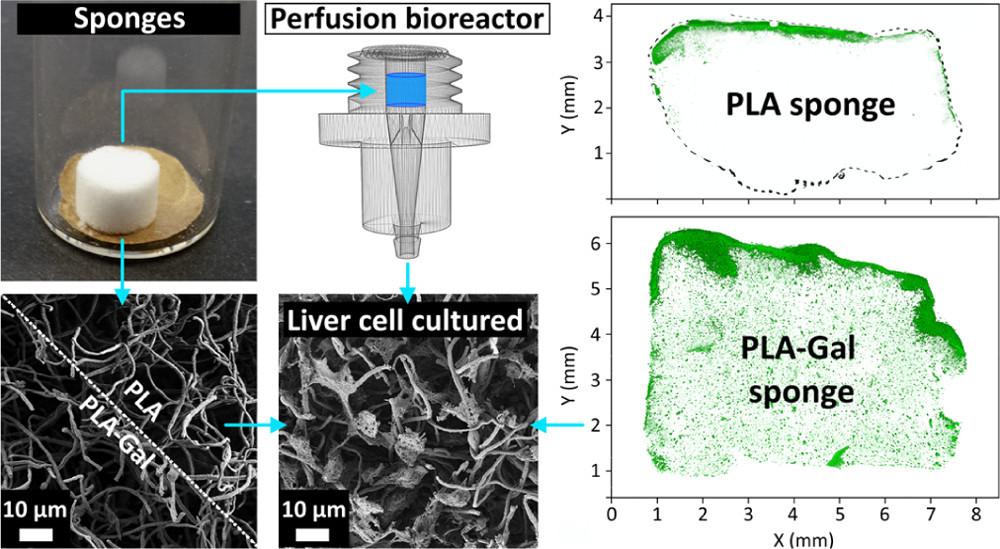
A major challenge in tissue engineering and artificial scaffolding is to combine easily tunable scaffolds biomimicking the extracellular matrix of native organs with delivery-controlled cell culturing to create fully cellularized, large artificial 3D scaffolds. Aiming at bioartificial liver construction, we present our research using galactose-functionalized, ultraporous polylactide 3D nanofiber sponges fabricated out of electrospun fibers. Sponge biomodification by blend galactosylation and in-solution coating is performed, respectively, using a polylactide-galactose carrier-copolymer that promotes cell delivery and features a pronounced autofluorescence. It allows us to verify the galactosylation success, evaluate its quality, and record dye-free, high-resolution images of the sponge network using confocal laser scanning microscopy. The galactose carrier and its impact on scaffold cellularization is validated in benchmark to several reference systems. Verification of the human hepatic cell asialoglycoprotein receptor presence and galactose interaction in culture is performed by Cu2+ receptor-blocking experiments. The culture results are extensively investigated in and ex situ to trace and quantify the cell culture progress, cell activity, and viability at different culture stages. Bioreactor cultivation of sponges reveals that the galactose carrier does not only facilitate cell adhesion but also enhances cellular distribution throughout the scaffold. The promising 3D culture results allow us to move forward to create mature in vitro liver model research systems. The elaboration into ex vivo testing platforms could help judging native cell material interactions with drugs or therapeutics, without the need of direct human or animal testing.

